Executive summary
A randomized, double-blind, sham-controlled, crossover study in healthy older adults showed that a single 20-minute session with the Vielight Neuro Gamma (810 nm LEDs pulsed at 40 Hz) increased alpha/beta/gamma power, decreased delta/theta, and significantly altered functional connectivity and graph-theory network measures (clustering, characteristic path length, local & global efficiency) relative to sham. These findings constitute non-invasive neuromodulation of human brain activity published in a Nature Portfolio journal (Scientific Reports).
Read the full publication here: Nature
As of Aug 20, 2025, the Vielight Neuro is involved in the only high-level journal publication demonstrating EEG neuromodulation with a commercially available, wearable LED brain PBM device.
Background: why test tPBM on neural oscillations?
Transcranial photobiomodulation (tPBM) uses low-level red/NIR light to modulate bioenergetic and signaling pathways (e.g., effects on the mitochondrial ETC and cytochrome-c oxidase), with accumulating human evidence in neurology and neurodegeneration. Yet, until this work, direct electrophysiological modulation in humans under rigorous controls had been limited. The study explicitly targeted the default mode network (DMN) with 40 Hz (gamma-range) pulsing, hypothesizing measurable EEG changes.
Methods at a glance

Figure 1. Schematic diagram of study design
-
Design: randomized, double-blind, sham-controlled, crossover; eyes-closed rest EEG recorded 10 min pre and 10 min post each session (active vs. sham), ≥1-week washout.
-
Participants: n = 20 healthy older adults (61–74 y).
-
Device & targeting: Vielight Neuro Gamma (wearable LEDs + intranasal emitter) delivering 810 nm, 40 Hz, 50% duty cycle, 20 min total, with modules positioned to stimulate DMN subdivisions (vmPFC via intranasal, dmPFC, PCC/precuneus, lateral parietal). Locations correspond to FPz, Cz, T3, T4 in the 10–20 montage.
-
Dosimetry summary: approximate beam spot ~1 cm²; power density transcranial anterior LED ~100 mW cm⁻²; intranasal ~25 mW cm⁻²; session energy density ~240 J cm⁻².
-
EEG analytics: spectral power (delta 1–3, theta 4–7, alpha 8–14, beta 14–30, gamma 30–50 Hz); functional connectivity via weighted phase-lag index (wPLI); graph-theory metrics—clustering coefficient (segregation), characteristic path length (integration), local and global efficiency.
Results
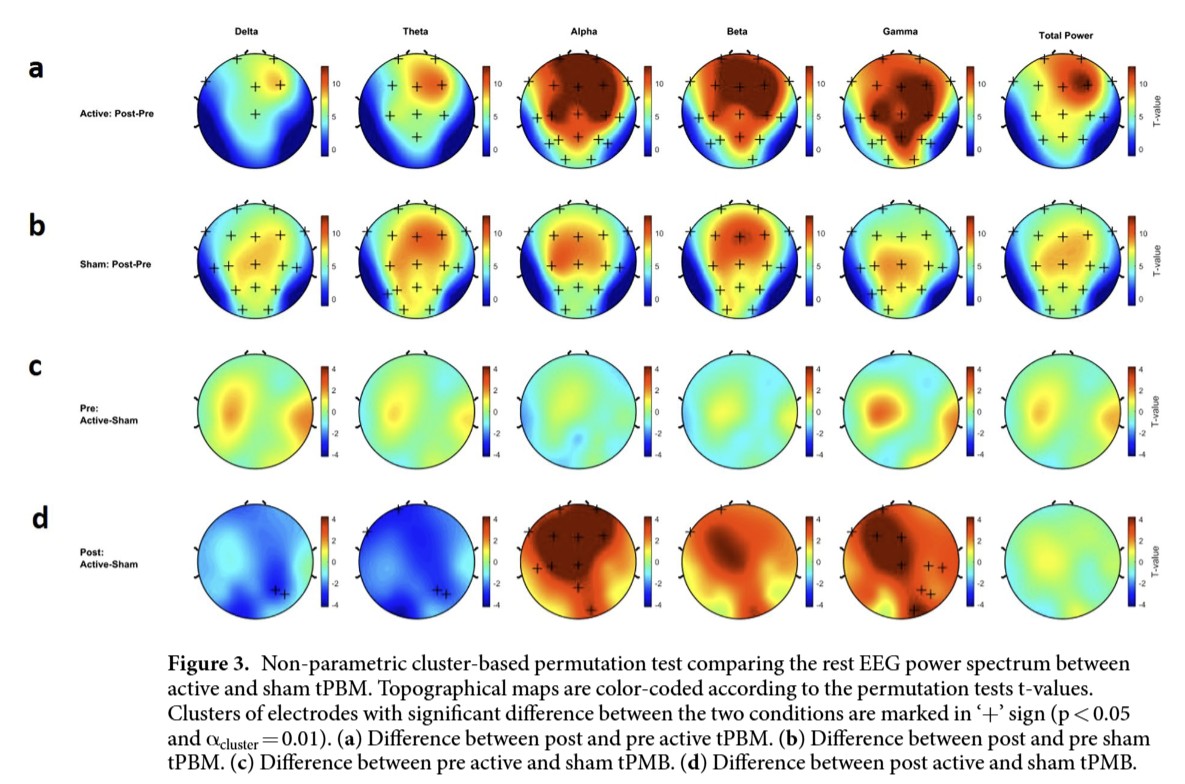
Figure 3. Non-parametric cluster-based permutation test comparing the rest EEG power spectrum betweenactive and sham tPBM
Power spectrum (active vs. sham)
Relative to sham, active 40 Hz, 810 nm tPBM produced a down-shift of low-frequency power and up-shift of higher-frequency power:
-
Delta ↓ (t = −3.513, p < 0.01), Theta ↓ (t = −3.736, p < 0.01)
-
Alpha ↑ (t = 4.455, p < 0.01), Beta ↑ (t = 3.221, p < 0.01), Gamma ↑ (t = 2.658, p < 0.01)
Directional effects: suppression of delta/theta increase seen with sham and facilitation of alpha/beta/gamma power. Nature

Figure 4. Influence of tPBM on resting-state electroencephalography.
Network connectivity & synchrony (wPLI + graph metrics)
Active tPBM induced statistically significant changes in clustering, characteristic path length (CPL), and local/global efficiency across sparsity thresholds, with the strongest effects in the alpha band; no comparable changes occurred under sham. Global efficiency changes were also detected in alpha and gamma ranges. Together, these indicate reorganization of functional integration and segregation following a single session.
DMN-linked interpretation
Given the DMN-centric montage, the authors note that increases in alpha (posterior DMN) and gamma (mPFC) are consistent with literature connecting these rhythms to DMN function and attentional gating. The pattern suggests greater network organization post-stimulation.
Why this matters (scientific implications)
-
Mechanistic neuromodulation in humans: Under stringent controls, 40 Hz/810 nm non-invasively modulated oscillations and network topology—a benchmark for tPBM as a bona fide neuromodulatory modality (not merely hemodynamic or metabolic).
-
Rhythmicity matters: The pulsing at 40 Hz (gamma) likely contributed—aligning with inhibitory control, binding, and attention models. Parameterization (wavelength, pulse rate, montage) therefore becomes a tractable “design space” for targeted outcomes.
-
Network-level targets: Changes in clustering, CPL, efficiency offer quantitative endpoints beyond band power—useful for designing protocols that aim to normalize DMN dynamics (e.g., conditions characterized by alpha/gamma deficits and delta/theta excess).
Clinical implications of this study
1. Proof of Neuromodulation in Humans
-
The findings show that Vielight Neuro Gamma is capable of non-invasively modulating brain oscillations in real time.
-
The shift toward increased alpha, beta, and gamma activity with concurrent decreases in delta and theta indicates a move toward more alert, engaged, and efficient brain states, which are typically associated with cognition and attention.
-
This establishes that transcranial + intranasal PBM isn’t just delivering NIR light, it is measurably reorganizing brain networks.
2. Functional Network Optimization
-
The improvements in graph-theory measures (clustering, path length, efficiency) point toward enhanced brain network integration and communication.
-
These are the same network-level metrics that are impaired in neurodegenerative diseases (e.g., Alzheimer’s, Parkinson’s), traumatic brain injury, and cognitive aging.
-
The implication: if a single 20-minute session can shift networks in healthy older adults, repeated sessions may hold therapeutic potential in clinical populations.
3. Bridge Between Basic Science and Clinical Application
-
Published in a Nature Portfolio journal (Scientific Reports), this gives strong credibility to PBM as a neuromodulation modality-on par conceptually with TMS or tDCS, but non-invasive and low-risk.
-
This sets a scientific foundation for expanding trials into clinical populations with cognitive decline, mood disorders, TBI, or performance optimization needs.
4. Early Biomarker of Efficacy
-
EEG and connectivity changes are quantifiable biomarkers.
-
Having objective measures allows clinicians to track response to treatment and tailor protocols, which is critical for PBM to be accepted in mainstream neurology/psychiatry.
Clinical Implication (in short):
This study provides high-level evidence that a single Vielight Neuro Gamma session produces measurable neuromodulation in the aging human brain. The changes in oscillatory activity and network efficiency are highly relevant to conditions marked by disrupted brain rhythms and connectivity (e.g., dementia, TBI, depression). It positions PBM as a credible, non-invasive therapeutic candidate and justifies larger, longitudinal clinical trials.
Practical implications for neurofeedback
-
Actionable biomarkers: The ↑alpha/β/γ, ↓δ/θ signature can serve as pre-/post-session metrics or training setpoints, potentially shortening session times by pre-conditioning networks before operant training.
-
Network-aware protocols: Because integration/segregation measures changed, practitioners can track wPLI-derived connectivity and efficiency as secondary outcomes, aligning neurofeedback goals with network-level normalization rather than single-electrode metrics alone.
Implications for AI and closed-loop neuromodulation
-
Real-time controllers: EEG power and wPLI-based metrics evolve quickly and can be optimized by AI (e.g., reinforcement learning) to adapt pulse parameters (timing, duty cycle, emitter grouping) toward desired states.
-
Multimodal causal structure: New Nature-Portfolio work demonstrates directed coupling among EEG, hemodynamics (HbO), and metabolism (cytochrome-c-oxidase) following tPBM—providing rich supervisory signals for multimodal AI to learn causal control policies for closed-loop tPBM. Nature
Limitations
-
Network effects depend on sparsity thresholds and graph-construction choices; replication with pre-registered pipelines is warranted.
















